This page will be updated weekly with new information on COVID-19
COVID-19 SUMMARY
You can also find this page via tabs at the top of the homepage
This page will be updated weekly with new information on COVID-19
COVID-19 SUMMARY
You can also find this page via tabs at the top of the homepage
There’s so much information about the novel coronavirus COVID-19 on the internet that it is hard to keep up with the onslaught of information. We wanted to compile the ultimate list of reputable resources for clinical providers to access when they need to, at a moment’s time.
Since information on COVID-19 is rapidly changing, these resources are not specific papers or blog posts, but rather websites that are maintaining up to date information on epidemiology, pathophysiology, and management. Resources span all types, including website behemoths like the WHO and CDC, as well as twitter accounts for people who get their news from social media.
Since this is an ultimate list but we don’t pretend to have ultimate knowledge of all resources, please send us resources that we may have missed and we will add them on here! We also acknowledge that this list is U.S. centric because we are from the States, but we would love input on resources for other countries so we can make this a more international list.
Graphics Reuters – for the graphic enthusiasts out there
New York Times Coronavirus Maps – for the graphic enthusiasts out there
That’s it for now! Let us know if we missed any great resources!
Thank you for all the work that all of you do. Stay safe!
This list was compiled by Milana Bogorodskaya, Fatima Al-Dhaheri, and Ahmed Abdul Azim.
Learning about fungi is hard enough even for infectious disease fellows (Narrator: especially for infectious disease fellows). By the time you learn how to differentiate the yeasts from the molds, the fungi kingdom decides to throw you a curve ball: Enter the shape shifters into the game of fungi learning – the dimorphic fungi.
The Dimorphic fungi shape shift depending on the weather (literally). They exist as molds in the great outdoors (environmental temperatures) and yeasts in the great indoors (inside our bodies at body temperatures). Clinically, this also means you will see the yeast forms in a histopathology review of a tissue sample, and our friends in the microbiology lab can re-create the environmental factors to grow them out as mold forms in culture. So essentially, they also shape shift between the microbiology lab and the pathology department. (They are sneaky Fung(uy)i…).
If you haven’t read the first post on Histoplasma capsulatum, go read it now!
This is the 2nd post out of 6 and will focus on our second shapeshifter, Blastomyces dermatiditis.
CLICK HERE for a 2-page PDF handout of this information.

At 25°C-30°C (mold form): septate hyphae with short or long conidiophores where a pear-shaped conidia form at the apex of the conidiophore (has a lollipop-like appearance).
(Image A)

At 37°C (yeast form):
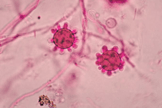
appear as yeast-like cells, thick walled and budding with a broad base
(Image B)
Your friendly Infectious disease doctors will alwaysask for tissue, and if classic broad-based budding yeast are appreciated in histopathology, that’s a slam dunk diagnosis! But we understand that is not always feasible, so in addition to clinical history + presentation, in order of importance:
Culture:
*Please alert the microbiology lab if you suspect Blastomycosis and are sending them cultures! (culture needs to be specially handled in the lab due to risk occupational transmission/infection (just like all dimorphic fungi covered in this review series).
Histopathology:
Antigen detection:
High sensitivity for urine detection (93%) in largest published evaluation(2) but low specificity (79%) due to cross reactivity with Histoplasmosis, Paracoccidioidomycosis and Talaromycosis (previously known as penicilliosis)
Serology:
Molecular methods:
Pulmonary disease
Disseminated disease
References:
1. Walsh TJ, Hayden RT, Larone DH. Larone’s medically important fungi, 6th edition, ASM press, 2018.
2. Saccente M and Woods GL. Clinical and laboratory update on blastomycosis. Clin Microbiol Rev. 2010;23:367–381.
3. Chapman SW, Dismukes WE, Proia LA, Bradsher RW, Pappas PG, Threlkeld MG, et al. Clinical practice guidelines for the management of blastomycosis: 2008 update by the Infectious Diseases Society of America. Clin Infect Dis. 2008 Jun 15;46(12):1801-12
4. Salzer HJF, Burchard G, Cornely OA, et al. Diagnosis and management of systemic endemic mycoses causing pulmonary disease. Respiration; 2018;96:283–301.
5. Pappas PG, Pottage JC, Powderly WG, et al. Blastomycosis in patients with the acquired immunodeficiency syndrome. Ann Intern Med;1992:116
Learning about fungi is hard enough even for infectious disease fellows (Narrator: especially for infectious disease fellows). By the time you learn how to differentiate the yeasts from the molds, the fungi kingdom decides to throw you a curve ball: Enter the shape shifters into the game of fungi learning – the dimorphic fungi.
The Dimorphic fungi shape shift depending on the weather (literally). They exist as molds in the great outdoors (environmental temperatures) and yeasts in the great indoors (inside our bodies at body temperatures). Clinically, this also means you will see the yeast forms in a histopathology review of a tissue sample, and our friends in the microbiology lab can re-create the environmental factors to grow them out as mold forms in culture. So essentially, they also shape shift between the microbiology lab and the pathology department. (They are sneaky Fung(uy)i…)
This is the first post out of 6 and will focus on our first shapeshifter, Histoplasma capsulatum.
CLICK HERE for a 2-page PDF handout of this information.

1. Culture:
2. Histopathology:
3. Antigen detection:
4. Serology:
5. Molecular methods:
| Clinical presentation | Mild/Moderate | Moderate/Severe | Chronic |
| Pulmonary | <4weeks: none >4weeks: itraconazole for 6-12 months | Lipid amphotericin B for 1-2 weeks followed by itraconazole for 12 weeks | Itraconazole for 12 months |
| Disseminated | Itraconazole for 12 months | Lipid amphotericin B for 1-2 weeks followed by itraconazole for 12 months | N/A |
References:
1. Climate change: the role of the infectious disease community. Lancet Infect Dis. 2017; 17:1219.
2. Greer A, Ng V, and Fisman D. Climate change and infectious diseases in North America: the road ahead. CMAJ. 2008; 178:715–722.
3. Walsh, TJ, Hayden, RT, and Larone, DH. Larone’s medically important fungi, 6th edition, ASM press, 2018.
4. Queiroz-Telles F, Fahal AH, Falci DR, et al. Neglected endemic mycoses. Lancet Infect Dis. 2017;17:e367–e377.
5. Azar MM and Hage CA. Laboratory Diagnostics for Histoplasmosis. J Clin Microbiol. 2017; 55:1612–1620.
6. Hage CA, Azar MM, Bahr N, Loyd J, and Wheat LJ. Histoplasmosis: up-to-date evidence-based approach to diagnosis and management. Semin Respir Crit Care Med. 2015; 36:729–745.
7. Kauffman CA. Histoplasmosis: a clinical and laboratory update. Clin Microbiol Rev. 2007;20:115–132.
8. Hage CA, Ribes JA, Wengenack NL, et al. A multicenter evaluation of tests for diagnosis of histoplasmosis. Clin Infect Dis. 2011;53:448–454.
9. Wheat LJ and Kauffman CA. Histoplasmosis. Infect Dis Clin North Am. 2003;17:1–19.
10. Swartzentruber S, Rhodes L, Kurkjian K, et al. Diagnosis of acute pulmonary histoplasmosis by antigen detection. Clin Infect Dis. 2009; 49:1878–1882.
11. Saccente M and Woods GL. Clinical and laboratory update on blastomycosis. Clin Microbiol Rev. 2010;23:367–381.
12. Wheat LJ, Freifeld AG, Kleiman MB, et al; Infectious Diseases Society of America. Clinical practice guidelines for the management of patients with histoplasmosis: 2007 update by the Infectious Diseases Society of America. Clin Infect Dis. 2007;45:807–825.

Learning about fungi is hard enough even for infectious disease fellows (Narrator: especially for infectious disease fellows). By the time you learn how to differentiate the yeasts from the molds, the fungi kingdom decides to throw you a curve ball: Enter the shape shifters into the game of fungi learning – the dimorphic fungi.
The Dimorphic fungi shape shift depending on the weather (literally). They exist as molds in the great outdoors (environmental temperatures) and yeasts in the great indoors (inside our bodies at body temperatures). Clinically, this also means you will see the yeast forms in a histopathology review of a tissue sample, and our friends in the microbiology lab can re-create the environmental factors to grow them out as mold forms in culture. So essentially, they also shape shift between the microbiology lab and the pathology department. (They are sneaky Fung(uy)i…)
Some of the clinically relevant dimorphic fungi have a predilection for geographical location (endemic mycoses), and therefore are very popular in board exams to the dismay (or joy, after this review series?) of medical trainees.
#ClimateChangeIsReal isn’t just pertinent in the political arena, but also for these endemic fungi. The grave consequences of climate change might change and expand the geographical distribution(1,2) of these fungi and therefore result in more catch-up learning on our end. This is almost akin to learning the constant re-classification and re-naming of the fungi kingdom (thanks, no thanks taxonomists…)
In this review series, I will go over the endemic fungi in a ‘high yield’ approach that will hopefully be pertinent for both shelf exams/boards and clinical practice.
I’ve also purposefully made it a two-pager/per fungi review (or 1 pager if you print it double-sided, #SaveTheTrees). We will be providing PDF links with every Fungi review. This will be an easy reference for a pocketbook, handouts to print to teach your medical students or if you want to flex your knowledge of endemic fungi during rounds (All win-win-win situations!)
The profile of each shape shifter will be released every Friday in the spirit of #FungalFriday. The dimorphic fungi that will be covered during the #ShapeShifterSeries include:
Our First Shape shifter in the series to be released this coming #FungalFriday will be Histoplasmosis, aka the Ohio valley disease/Cave disease. What does Ohio or caves for the matter have to do with this Fungus? Find out more this coming Friday!
Fatima Al Dhaheri, MBBS
The Fung(uy)i squad
References:
1. The Lancet Infectious Diseases. Climate change: the role of the infectious disease community. Lancet Infect Dis. 2017;17:1219.
2. Greer A, Ng V, Fisman D. Climate change and infectious diseases in North America: the road ahead. CMAJ. 2008;178:715–722.

[This post was written by Ahmed Abdul Azim, a senior infectious disease fellow at Beth Israel Deaconess Medical Center]
During the fall and winter season, you are likely to see a few cases of viral meningitis. Even though viral encephalitis is less common, it is important to try to differentiate these clinical entities as a clinician, since they carry different prognoses. (The bulk of this review is adapted from Mandell, Douglas and Bennett’s principles and practice of infectious diseases)1
Before we go any further, let’s briefly review cerebral spinal fluid findings on lumbar puncture for different syndromes:
| WBC(cells/mm3) | Primary cells | Glucose(mg/dL) | Protein(mg/dL) | |
| Viral | 50-1000 | Lymphocytic | >45 | <200 |
| Bacterial | 1000-5000 | Neutrophilic | <40 | 100-500 |
| Mycobacterial | 50-500 | Lymphocytic | <45 | 50-300 |
| Cryptococcal/fungal | 20-500 | Lymphocytic | <40 | >45 |
Important points to consider:
· Bacterial meningitis: 10% of cases have a lymphocyte predominant CSF cell analysis
· WNV encephalitis: over a 1/3 of patients with WNV encephalitis had neutrophil predominant CSF pleocytosis
· Enteroviruses: CSF analysis done early in illness course may yield neutrophil predominant pleocytosis in 2/3 of cases – generally will convert to lymphocytic predominant if repeated in 12-24 hours.
Take home point: always interpret CSF within the clinical context in front of you!
Both syndromes often present with a triad of2:
(1) FEVER
(2) HEADACHE and
(3) ALTERED MENTAL STATUS
However, the trick is to explore the history and signs further. Epidemiological clues include:
Patients with viral encephalitis: tend to have diffuse cerebral cortex involvement with abnormal cerebral function
– Symptoms: altered mental status, motor/sensory deficits, altered behavior and/or personality changes, speech and/or movement disorders
Patients with viral meningitis: DO NOT have diffuse cerebral cortex involvement → cerebral function IS INTACT
– Symptoms: headache, lethargy, neck stiffness/pain
Patients with meningoencephalitis: tend to have a combination of meningitis and encephalitis symptoms
Regardless, if a patient has symptoms and/or signs of meningitis or encephalitis, a lumbar puncture can be helpful.
Overall, most cases of aspectic meningitis syndromes are caused by viruses
1. Enteroviruses (e.g. Coxsackie, echovirus, other non-polio enteroviruses) – by far the most common cause of viral meningitis/aseptic meningitis3
2. Herpes virus simplex viral meningitis – usually caused by HSV-2 >> HSV-18
3. West Nile Virus – more likely to cause an encephalitis syndrome. Yet, may present with aseptic meningitis or asymmetrical flaccid paralysis10
A cause is identified in approximately 36-63% of cases10,11
Causes of encephalitis (Most common to least common in US study of patients that met criteria for encephalitis)12:
*In a study of HIV uninfected patients, viruses caused up to 38% of cases, followed by bacterial pathogens at 33%, Lyme disease at 7%, and fungi at 7%. Syphilis was identified as the culprit in 5% of cases, and mycobacterial infections at 5%, while prion disease was responsible for 3% of cases of encephalitis11
1. HSV encephalitis: most common cause of encephalitis in the US (1/250,000 population annually). HSV-1 accounts for greater than 90% of HSV encephalitis in adults13. Fewer than 6% of CSF PCR cases had a “normal” neurological exam14.
2. West Nile Virus encephalitis: transmitted via a mosquito (vector) bite, currently the most common cause of epidemic viral encephalitis nationally19
So you are the house officer encountering a patient with 1-2 weeks of progressively worsening fevers, headaches and severe behavioral changes or depressed mental status: what do you do next?
As a standard work up for likely encephalitis in the United States, CSF studies should include1:
Imaging in encephalitis: Magnetic resonance imaging (MRI) of the brain is more sensitive than computed tomography (CT)15. Unless contraindicated, all patients with encephalitis should undergo MR imaging.
1. Mandell, Douglas and Bennett’s principles and practice of infectious diseases (8th ed. 2015 / Philadelphia, PA : Elsevier)
2. Whitley RJ, and Gnann JW: Viral encephalitis: familiar infections and emerging pathogens. Lancet 2002; 359: pp. 507-513
3. Connolly KJ, and Hammer SM: The acute aseptic meningitis syndrome. Infect Dis Clin North Am 1990; 4: pp. 599-622
4. Gomez B, Mintegi S, Rubio MC, et al: Clinical and analytical characteristics and short-term evolution of enteroviral meningitis in young infants presenting with fever without source. Pediatr Emerg Care 2012; 28: pp. 518-523
5. Rotbart HA: Diagnosis of enteroviral meningitis with the polymerase chain reaction. J Pediatr 1990; 117: pp. 85-89
6. Sawyer MH, Holland D, and Aintablian N: Diagnosis of enteroviral central nervous system infection by polymerase chain reaction during a large community outbreak. Pediatr Infect Dis J 1994; 13: pp. 177-182
7. Ahmed A, Brito F, Goto C, et al: Clinical utility of polymerase chain reaction for diagnosis of enteroviral meningitis in infancy. J Pediatr 1997; 131: pp. 393-397
8. Shalabi M, and Whitley RJ: Recurrent benign lymphocytic meningitis. Clin Infect Dis 2006; 43: pp. 1194-1197
9. Corey L, and Spear PG: Infections with herpes simplex viruses (2). N Engl J Med 1986; 314: pp. 749-757
10. Kupila L, Vuorinen T, Vainionpaa R, et al: Etiology of aseptic meningitis and encephalitis in an adult population. Neurology 2006; 66: pp. 75-80
11. Tan K, Patel S, Gandhi N, et al: Burden of neuroinfectious diseases on the neurology service in a tertiary care center. Neurology 2008; 71: pp. 1160-1166
12. Glaser CA, Gilliam S, Schnurr D, et al: In search of encephalitis etiologies: diagnostic challenges in the California Encephalitis Project, 1998-2000. Clin Infect Dis 2003; 36: pp. 731-742
13. Tyler KL: Herpes simplex virus infections of the central nervous system: encephalitis and meningitis, including Mollaret’s. Herpes 2004; 11: pp. 57A-64A
14. Raschilas F, Wolff M, Delatour F, et al: Outcome of and prognostic factors for herpes simplex encephalitis in adult patients: results of a multicenter study. Clin Infect Dis 2002; 35: pp. 254-26
15. Domingues RB, Tsanaclis AM, Pannuti CS, et al: Evaluation of the range of clinical presentations of herpes simplex encephalitis by using polymerase chain reaction assay of cerebrospinal fluid samples. Clin Infect Dis 1997; 25: pp. 86-91
16. Whitley RJ, Alford CA, Hirsch MS, et al: Vidarabine versus acyclovir therapy in herpes simplex encephalitis. N Engl J Med 1986; 314: pp. 144-149
17. Lakeman FD, and Whitley RJ: Diagnosis of herpes simplex encephalitis: application of polymerase chain reaction to cerebrospinal fluid from brain-biopsied patients and correlation with disease. National Institute of Allergy and Infectious Diseases Collaborative Antiviral Study Group. J Infect Dis 1995; 171: pp. 857-86
18. Whitley RJ, Alford CA, Hirsch MS, et al: Factors indicative of outcome in a comparative trial of acyclovir and vidarabine for biopsy-proven herpes simplex encephalitis. Infection 1987; 15: pp. S3-S8
19. Kramer LD, Li J, and Shi PY: West Nile virus. Lancet Neurol 2007; 6: pp. 171-181
20. Watson JT, Pertel PE, Jones RC, et al: Clinical characteristics and functional outcomes of West Nile Fever. Ann Intern Med 2004; 141: pp. 360-365
21. Sejvar JJ, Haddad MB, Tierney BC, et al: Neurologic manifestations and outcome of West Nile virus infection. JAMA 2003; 290: pp. 511-515
22. Jean CM, Honarmand S, Louie JK, et al: Risk factors for West Nile virus neuroinvasive disease, California, 2005. Emerg Infect Dis 2007; 13: pp. 1918-1920
23. Shi PY, and Wong SJ: Serologic diagnosis of West Nile virus infection. Expert Rev Mol Diagn 2003; 3: pp. 733-741
24. Garcia MN, Hause AM, Walker CM, et al: Evaluation of prolonged fatigue post-West Nile virus infection and association of fatigue with elevated antiviral and proinflammatory cytokines. Viral Immunol 2014; 27: pp. 327-333
25. Gilden DH, Mahalingam R, Cohrs RJ, et al: Herpesvirus infections of the nervous system. Nat Clin Pract Neurol 2007; 3: pp. 82-94
The ATS and IDSA recently released the much-anticipated update to the community-acquired pneumonia (CAP) guidelines. The previous version was published back in 2007 and the new guidelines have included some major changes. Here is a rundown of all those changes that you need to know.
1. Health care associated pneumonia (HCAP) no longer exists
HCAP was an entity created with the 2007 CAP guidelines. It encompassed non-hospital acquired pneumonia in patients who had recent contact with the healthcare system. The recommendation was to treat HCAP with empiric broad-spectrum antibiotic therapy against methicillin-resistant Staphylococcus aureus (MRSA) and Pseudomonas aeruginosa (PsA). With this strategy however, we were over-treating a lot of people. This study found that while 30% of all patients hospitalized for CAP received empiric anti-MRSA treatment, only 0.7% of all patients had MRSA pneumonia.
In the new guidelines, HCAP no longer exists. Instead, the guidelines emphasize assessment of risk factors for pathogens such as MRSA and PsA.

2. Treatment is now based on severity of the pneumonia rather than the location of the admitted patient
Prior guidelines differentiated antibiotic recommendations based on patient triage to the floor or the intensive care unit. In the new guidelines, treatment recommendations are based on the severity of the pneumonia, based on a list of criteria:

3. Only obtain blood cultures in severe CAP or if risk factors for MRSA and/or PsA are present
The new guidelines focus on cost-effective use of diagnostic tests.
Outpatient setting: recommend against any diagnostic testing (except for a chest X-ray)
Inpatient non-severe pneumonia: recommend blood cultures and sputum gram stain/culture ONLY if risk factors for MRSA and/or PsA are present
Inpatient severe pneumonia: recommend blood cultures, sputum gram stain/culture, Streptococcus pneumoniae urine antigen, and Legionella urine antigen and PCR/culture
*Legionella diagnostic tests are also recommended in times of an outbreak
These recommendations are based on literature demonstrating that:
4. Procalcitonin should NOT be used in the diagnosis of CAP
Procalcitonin is not a reliable marker for diagnosis of bacterial infections; it has roughly 65-75% sensitivity for detecting bacterial pneumonia8. Consequently, the risk of not treating bacterial CAP due to a low procalcitonin level can lead to poor outcomes. Although there is data to support use of procalcitonin in determining the duration of antibiotics in CAP9,10, the guidelines recommend use only in situations where duration exceeds the recommended 5-7 days.
5. The guidelines recommend use of the Pneumonia Severity Index (PSI) over the CURB-65 for determining need for admission
The argument from the guideline authors is that there is more literature in support for PSI in accurately predicting mortality rather than the CURB-65 score11-14. However, PSI incorporates data that may not be available in all circumstances, and certainly will not be available to the outpatient clinician who is trying to decide whether to admit a patient or not (such as pH, which can only be obtained from an arterial blood gas). So, although PSI may be recommended for use in the emergency department, the CURB-65 will likely remain in use, especially due to its efficiency in the outpatient setting.
6. Algorithm for CAP antibiotic treatment
The meat of the guidelines is the treatment regimens – and there are quite a few changes.
You may be thinking – “wait, amoxicillin doesn’t even cover atypical pathogens (i.e. Mycoplasma pneumoniae and Legionella pneumophila)!” This is true. But studies have shown that in otherwise-healthy patients, there was no difference in outcomes among those who received amoxicillin vs. an antibiotic that treats atypical organisms16. Exactly why that is remains unclear, but could be because healthy individuals clear the infection on their own or because the majority of these pneumonias are actually due to a virus, so they would improve with or without any antibiotics 5.


7. Treat anaerobes only in cases with suspected or proven lung abscess and/or empyema
Empiric treatment of anaerobes in aspiration pneumonia remains controversial, but the new guidelines recommend only treating anaerobes if there is suspicion for or a proven lung abscess and/or empyema.
8. Continue antibiotics for at least 48 hours in patients who are diagnosed with influenza pneumonia
This recommendation is based off the data that influenza infection predisposes to subsequent bacterial superinfections17 and a patient could have both a viral and a bacterial pneumonia at the same time. The guidelines state that if there is significant clinical improvement in 48 hours and no evidence to suggest a superimposed bacterial pneumonia, antibiotics can be discontinued at that time.
9. Duration of antibiotics is based on clinical improvement (but should be a minimum of 5 days)
Gone are the days of prespecified number of days for antibiotic duration. Instead, monitor the patient for signs of clinical improvement.
10. Do not use corticosteroids as adjunctive treatment and do not obtain routine follow up chest X-rays
These were not necessarily strategies that I employed prior to the publication of these guidelines, and corticosteroid use in CAP is controversial, but at this time, there is no strong data to support either of these adjunctive management strategies in patients with CAP.
References:
1. Metlay JP, Waterer GW, Long AC, et al. Diagnosis and Treatment of Adults with Community-acquired Pneumonia. An Official Clinical Practice Guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Resp Crit Care Med. 2019; 200(7):e45-e67.
2. Self WH, Wunderink RG, Williams DJ, et al. Staphylococcus aureus Community-acquired Pneumonia: Prevalence, Clinical Characteristics, and Outcomes. Clin Infect Dis. 2016; 63(3):300-309.
3. Chalasani NP, Valdecanas MA, Gopal AK, McGowan JE Jr, and Jurado RL. Clinical utility of blood cultures in adult patients with community-acquired pneumonia without defined underlying risks. Chest. 1995; 108(4):932-936.
4. Corbo J, Friedman B, Bijur P, and Gallagher EJ. Limited usefulness of initial blood cultures in community acquired pneumonia. Emerg Med J. 2004; 21(4):446-448.
5. Jain S, Self WH, Wunderink RG, et al. Community-Acquired Pneumonia Requiring Hospitalization among U.S. Adults. New Eng J Med. 2015. 373:415-427.
6. Lee JH and Kim YH. Predictive factors of true bacteremia and the clinical utility of blood cultures as a prognostic tool in patients with community-onset pneumonia. Medicine (Baltimore). 2016; 95(41):e5058.
7. Waterer GW and Wunderink RG. The influence of the severity of community-acquired pneumonia on the usefulness of blood cultures. Respir Med. 2001; 95(1):78-82.
8. Self WH, Balk RA, Grijalva CG, et al. Procalcitonin as a Marker of Etiology in Adults Hospitalized With Community-Acquired Pneumonia. Clin Infect Dis. 2017;65(2):183-190.
9. Schuetz P, Wirz Y, Sager R, et al. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. Cochrane Database Syst Rev. 2017; 10:CD007498.
10. Schuetz P, Wirz Y, Sager R, et al. Effect of procalcitonin-guided antibiotic treatment on mortality in acute respiratory infections: a patient level meta-analysis. Lancet Infect Dis. 2018;18(1):95-107.
11. Aujesky D, Auble TE, Yealy DM, et al. Prospective comparison of three validated prediction rules for prognosis in community-acquired pneumonia. Am J Med. 2005; 118(4):384-392.
12. Marrie TJ, Lau CY, Wheeler SL, Wong CJ, Vandervoort MK, and Feagan BG. A controlled trial of a critical pathway for treatment of community-acquired pneumonia. CAPITAL Study Investigators. Community-Acquired Pneumonia Intervention Trial Assessing Levofloxacin. JAMA. 2000; 283(6):749-755.
13. Carratala J, Fernandez-Sabe N, Ortega L, et al. Outpatient care compared with hospitalization for community-acquired pneumonia: a randomized trial in low-risk patients. Ann Intern Med. 2005;142(3):165-172.
14. Renaud B, Coma E, Labarere J, et al. Routine use of the Pneumonia Severity Index for guiding the site-of-treatment decision of patients with pneumonia in the emergency department: a multicenter, prospective, observational, controlled cohort study. Clin Infect Dis. 2007;441(1):41-49.
15. Blondeau JM and Theriault N. Application of the Formula for Rational Antimicrobial Therapy (FRAT) to Community-Acquired Pneumonia. J Infect Dis Ther. 2017;5:313.
16. Postma DW, van Werkhoven CH, van Elden LJR, et al. Antibiotic Treatment Strategies for Community-acquired Pneumonia in Adults. New Eng J Med. 2015;372:1312-1323.
17. Metersky ML, Masterton RG, Lode H, File TM Jr, and Babinchak T. Epidemiology, microbiology, and treatment considerations for bacterial pneumonia complicating influenza. Int J Infect Dis. 2012;16(5):e321-331.
This post is the last in a three-part series covering the management of beta-lactam allergies. Part 1 explained the enormous impact that penicillin allergies have on patient outcomes, while Part 2 discussed the different types of allergic reactions and the potential (or lack thereof) for beta-lactam allergy cross reactivity. This last post will cover the methods used to assess beta-lactam allergies. Let’s jump right in!
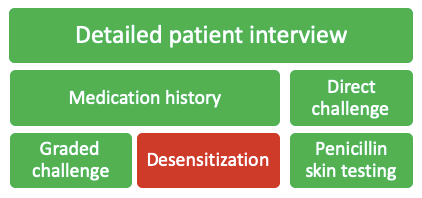
There are a variety of strategies that can be used to assess a patient’s beta-lactam allergy, each having their own place in the allergy assessment algorithm. The following will be detailed in this post:
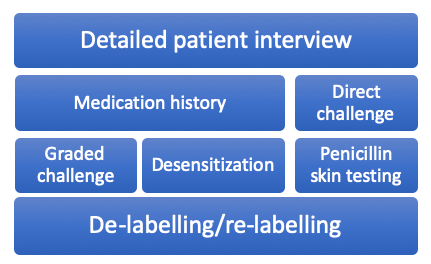
Detailed Patient Interview
Far and away the most important step in assessing a patient’s beta-lactam allergy is a detailed patient interview. An allergy evaluation is recommended by many of the top health organizations in the country, including the Center for Disease Control and Prevention (CDC), National Quality Forum, Infectious Diseases Society of America (IDSA), American Board of Internal Medicine (ABIM), and the American Academy of Allergy, Asthma & Immunology (AAAAI).1 Just a minute or two of questioning the patient can yield an entirely different story than the allergy history in the medical chart. Some common questions I bring up with patients include:
You can develop your own arsenal of questions to ask patients, but the important part is to talk to them. No further strategies are needed if you can rule out the documented allergy just from a 90-second conversation.
Medication History
The other piece that is absolutely necessary before proceeding is looking through the patient’s medication history yourself. If a patient with a documented penicillin allergy received ceftriaxone without issue on an admission last year, you can go ahead and give full-dose ceftriaxone during this admission if needed. The patient interview and medication history review can rule out >50% of documented allergies in my experience. In these situations, you can skip directly to the last section of this post: allergy re-labelling.
Direct Challenge

In patients with a very low probability of allergic reaction, a beta-lactam antibiotic can usually be given without pause. Situations where you can rule out an allergy based on patient interview or medication history can be “challenged” directly. This means giving the full dose of the preferred antibiotic and monitoring for any adverse effects. Some institutions also give a direct oral amoxicillin challenge with 250-500 mg of amoxicillin once prior to the intended beta-lactam initiation. If the patient can tolerate amoxicillin, any penicillin antibiotic can be given in the future without fear of experiencing an IgE-mediated reaction.
Graded Challenge
When you are not able to completely rule out an allergic reaction, a graded challenge is often the next logical step in hospitalized patients. Graded challenges are used when there is a low probability of an allergic reaction, but there is still a degree of discomfort giving the entire dose up front. In general, 10% of the full dose is given, the patient is monitored closely for 30 minutes, and then the full dose is given if no issues arise. If the patient tolerates these doses, you can rule out immediate hypersensitivity reactions and document the tolerance in the medical record, which will be discussed at the end of this post.
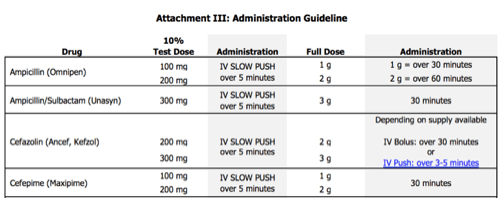
Desensitization

In patients who have confirmed or a high probability of severe IgE-mediated reactions to beta-lactams, but a beta-lactam is necessary for treatment, desensitization can be used. The desensitization procedure usually involves at least 12 doses of escalating concentrations of the required medication. This procedure requires incredibly close monitoring, which at most hospitals requires admission to the intensive care unit for administration. If a patient is able to tolerate desensitization, the patient must then begin regularly scheduled doses of the beta-lactam immediately upon the protocol completion. If doses are missed, the patient must be desensitized again. Desensitization does not rule out the allergy. The patient is still considered allergic to that agent, but can tolerate the medication for the course required in that instance.
Penicillin Skin Testing
Penicillin (PCN) skin testing has increased in popularity recently due to its relative ease of use and efficacy at ruling out IgE-mediated allergic reactions. In addition to rescue medications that should be handy just in case (diphenhydramine, methylprednisolone, and epinephrine) the skin test consists of 4 elements:

Initially, a percutaneous puncture test is done on the patient’s forearm with each of the elements and if tolerated, an intradermal test of each is also performed. The entire process generally takes around 45-60 minutes to complete and offers a negative predictive value for penicillin allergies of ~99%.2 Debate has surrounded the cost (both time and materials for the procedure), but multiple studies have now shown penicillin skin testing to be a cost-saving venture.2-5
Penicillin skin testing seems like a no-brainer, carrying the lowest risk of the procedures discussed thus far and its low overall cost for the health system. But in many institutions, it’s unclear who will perform the testing when allergy consultation is not available. In a 2015 survey of 736 infectious diseases providers, 57% responded saying that they do not have local options for skin testing.6 Does your institution?
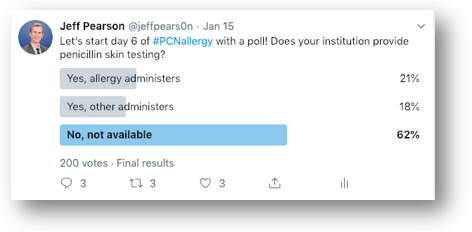
The people of Twitter have spoken and it resulted in similar responses, with 62% of respondents not having penicillin skin testing available at their institution. Previous studies have reported on the successes of penicillin skin testing performed by allergists,7-9 & many more antimicrobial stewardship programs,10 infectious diseases fellows/physicians,11 nurses,12 and pharmacists.13,14 If you’ve read this far into the post, you likely are interested in allergy skin testing, so I’d implore you to own the process if your institution doesn’t already have skin testing available! ALK provides some excellent instructional videos on their website to guide you through the testing process. Pharmacists aren’t licensed to perform skin testing in all 50 states, but they are in many of them, which this 2019 article did an admirable job exploring.15
Allergy re-labelling
The last fundamental step in navigating beta-lactam allergies is updating the patient’s allergy label. With all of the previous interventions, the allergy documentation can be further described in the medical record, with desensitization being the only intervention that does not rule out IgE-mediated reactions altogether.
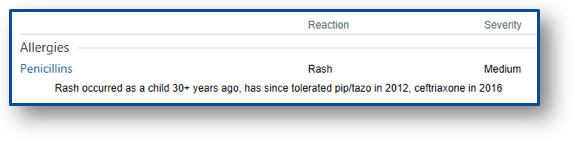
In an ideal world, inaccurate allergy labels should be removed from the medical record. Unfortunately, this practice often leads to redocumentation of the allergy at a later admission however.16 Many hospitals have integrated innovative ways to improve this repetitive cycle, as seen via providers’ personal experiences here, here, and here. For those without the tech support for any of this functionality though, the best thing to do is to document, document, document.
Summary
The majority of penicillin allergy labels do not belong to patients with true allergies and these unnecessary labels lead to worse patient outcomes. We should all strive for more accurate and detailed allergy documentation in our patients, which all starts with a patient interview. All of the interventions discussed above can be used to remove/relabel a beta-lactam allergy, with the exception of desensitization.
For those looking to learn more, I highly recommend a recent review published in JAMA that goes into further detail on penicillin allergies.17 Make sure to check out the supplementary material too, it has some super helpful resources, including a full allergy toolkit for penicillin skin testing and oral amoxicillin challenges!
Previous posts in this series:
A Rash of Beta-Lactam Allergies, Part 1: The Problem
A Rash of Beta-Lactam Allergies, Part 2: The Education
References
This post is the second in a three-part series covering the management of beta-lactam allergies, all to be released on FOAMid over the last few months of 2019. Part 1 explained the enormous impact that penicillin allergies have on patient outcomes. Today we’ll discuss the different types of allergic reactions and the potential for beta-lactam allergy cross reactivity. Let’s jump right in!
Types of Allergic Reactions
The most common way of grouping immune-mediated hypersensitivity reactions is through the Gell & Coombs classification method.2 Using this scheme, there are four types of allergic reaction:

Type I reactions are IgE-mediated reactions and commonly referred to as immediate-type hypersensitivity reactions, since they occur minutes to hours post-exposure to an allergen. Type I reactions include anaphylaxis, angioedema, hypotension, flushing, wheezing, hives, and urticaria.
Both types II and III reactions are IgG-mediated.
Type II reactions, or cytotoxic reactions, include hemolytic anemia, thrombocytopenia, and neutropenia.
Type III reactions are immune complex reactions, and include serum sickness, glomerulonephritis, and arthritis.
Last, but certainly not least, are type IV reactions, which are T-cell mediated.
Type IV reactions are commonly referred to as delayed hypersensitivity reactions, despite Types II, III, and IV all technically being delayed in nature by days to weeks post-exposure to an allergen. A maculopapular rash, interstitial nephritis, Stevens-Johnson Syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome are all considered type IV reactions.
Cross-Reactivity Risk
As discussed in the first post, many recorded antibiotic allergies are not true allergies. But when a patient does actually have a true penicillin allergy, what are the chances that the patient will have a similar reaction to other beta-lactams?
While we would generally avoid penicillins in this situation, other beta-lactams like cephalosporins and carbapenems could potentially be used. Previous studies of 10-25% cross-reactivity between cephalosporins and penicillins were primarily reported prior to 1982, when cephalosporin manufacturing processes were often contaminated with penicillin.12 Since then, the documented rate of cross-reactivity has dropped dramatically, shown in the table below.12

Cephalosporins
Side chain similarities don’t guarantee cross-reactivity, but they do increase the risk above the previously stated 2% threshold
But hold on, I thought the cause of beta-lactam allergies was the core beta-lactam ring that everyone remembers from their undergraduate years?

Not so fast. While this plays a part, more recent literature has shown that the R1 and R2 side chains also play a role in the allergy potential of cephalosporins. I have adopted and updated a table from an excellent 2008 review paper by Daryl DePestel and colleagues below.3
In this table, the 3s, 6s, and 7s stand for similar R1 or R2 side chains, as described in the cephalosporin skeleton molecule, also seen below.


There are a couple of important clinical points to note from this table. Probably most important for clinical practice is that cefazolin does not share side chains with any other beta-lactam agents. This can have huge consequences on the use of cefazolin in practice, especially when it comes to surgical site prophylaxis and the treatment of methicillin-susceptible Staphylococcus aureus infections, both situations that could use cefazolin as first line therapy.
And while aztreonam is known as a beta-lactam with limited cross-reactivity due to dissimilar side chains, it does actually share a side chain with ceftazidime and the more recently approved ceftolozane (marketed in combination with tazobactam).
Aztreonam & Carbapenems
Speaking of aztreonam, we’ve spent the majority of this post discussing cephalosporin cross-reactivity risk. Now let’s spend a bit of time reviewing the other agents defined in the initial table in this post: carbapenems and aztreonam. Cross-reactivity between these agents and penicillins is minimal, as seen by a number of studies published by an Italian group headed by Antonino Romano and Francesco Gaeta.4,7,9
In their 2013 analysis, they found no patients had an allergic reaction to carbapenems, despite all 204 patients having a well-demonstrated T-cell-mediated hypersensitivity reaction to other beta-lactams (mostly penicillin).7
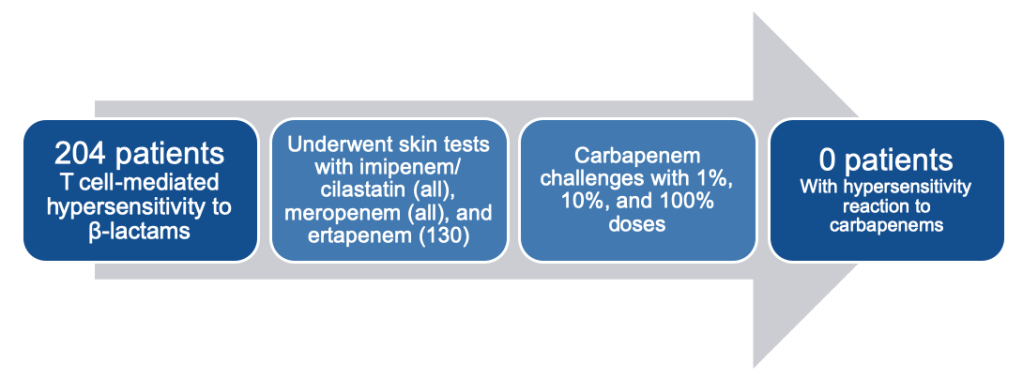
They went on to look at IgE-mediated hypersensitivity in their 2015 study, which found yet again no cases of hypersensitivity with either carbapenems OR aztreonam this time in a cohort of 212 patients with proven penicillin allergies.4

Then in 2016, they went back to T-cell-mediated hypersensitivity, examining 214 patients with proven reactions to penicillins and testing them against aztreonam. Once again, zero patients reacted to the aztreonam test doses or full dose.9

At this point, you may be questioning if the Italian group ever saw any reactions in their trial outcomes. The last study presented above that showed no reactions with aztreonam though tested more than just aztreonam. They also looked at cephalosporins and saw an 18.7% chance of positive skin testing with aminocephalosporins (cephalexin, cefadroxil, cefaclor).9 If you refer back to the previous cross-reactivity table, you can see that these three agents share a side chain with ampicillin and amoxicillin.

So while side chains play a key role in determining cross-reactivity among cephalosporins, we can be fairly confident that carbapenems and aztreonam are safe to administer in the majority of situations, especially when a non-severe penicillin allergy is documented. This will be covered in more detail in the next (and final) installment of “A Rash of Beta-Lactam Allergies,” coming to you soon!
Other posts in this series:
A Rash of Beta-Lactam Allergies, Part 1: The Problem
A Rash of Beta-Lactam Allergies, Part 3: The Solution
References
This post marks part 1 of a 3-part series covering the management of beta-lactam allergies, all to be released on FOAMid over the next couple of months.
With that, let’s jump right in!
Overview
A whopping 10% of the general population has a reported penicillin (PCN) allergy. But only 1-10% of these people have a true allergy when tested. This leaves us with about 0.1-1% of the general population with a true penicillin allergy.

Why is there such a discrepancy between reported allergies and true allergies? A lot of it comes from inaccurate allergy histories, like the patient with GI upset as a child, but the allergy listed as an “unknown reaction.” Or better yet, the patient whose mother had an allergy and thus everyone in the family has been given that scarlet letter in their medical record.

Another important and lesser known reason for the allergy discrepancy is that 78% of patients with immediate hypersensitivity to penicillin see their penicillin allergy fade after 10 years (from this 1981 study). So those adult patients with childhood reactions? The odds are that they aren’t still allergic decades later.
Why should we care?
When it comes to infectious diseases, beta-lactam antibiotics are often our first- and second-line options for treatment. A documented penicillin allergy can essentially knock a practitioner down to third-line treatment in some situations. In just highlighting a few common infections and organisms, look at how often beta-lactams are brought up:
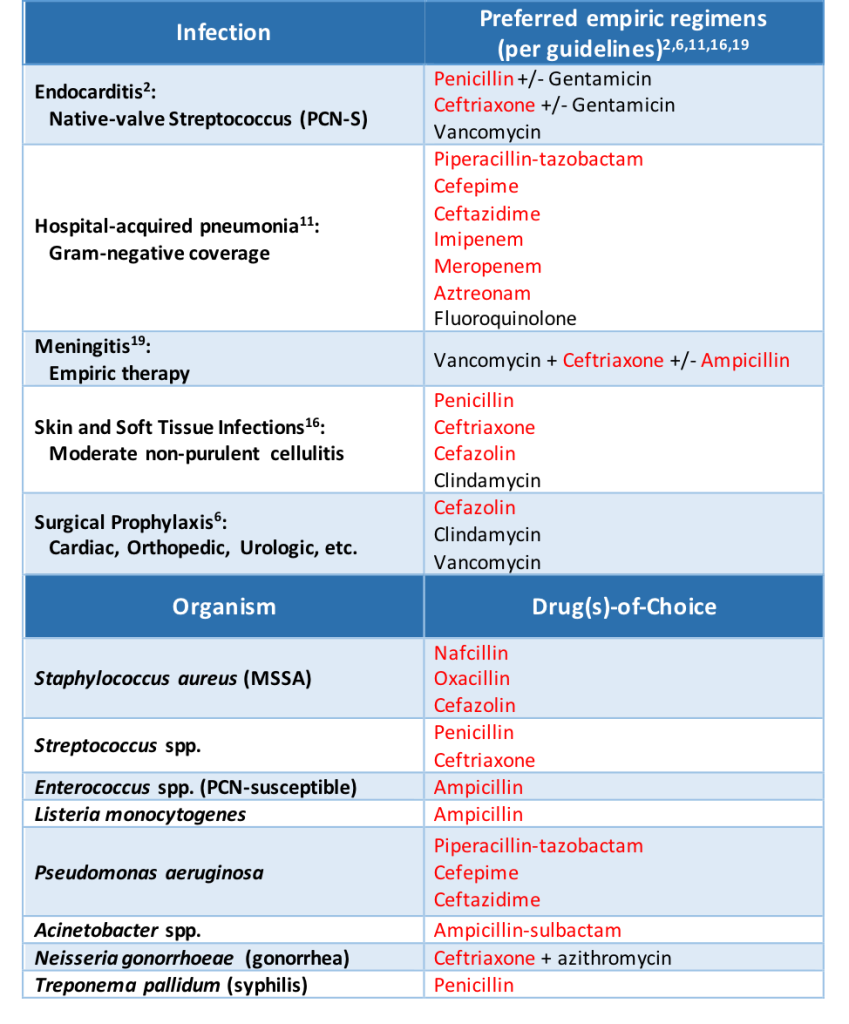
When a patient has a documented penicillin allergy, studies have proven that beta-lactam usage decreases while non-beta-lactam usage increases (Lee 2000, as well as half of the citations provided at the end of this post). And when beta-lactams are avoided, patients tend to do worse.
Impact on Patient Outcomes
The impact of a penicillin allergy is real and detrimental to our patients. Rather than bore you with paragraphs upon paragraphs detailing the many studies looking into this fact, here are some take-home points hyperlinked to the primary literature supporting the claims:
Penicillin allergy patients:
There is clear evidence that reported beta-lactam allergies pose a problem on the path to prescribing optimal treatment in infectious diseases. We can combat the issue however through education and assessment techniques.
More to come in parts 2 and 3 of “A Rash of Beta-Lactam Allergies”!
Other posts in this series:
A Rash of Beta-Lactam Allergies, Part 2: The Education
A Rash of Beta-Lactam Allergies, Part 3: The Solution
References